In this guide to understanding zinc as a commodity, we’ll explain why it’s valuable, how it’s produced, which countries produce the most zinc, and discuss what drives its price.
Interested in how zinc is traded? See our full guide, or if you want to get started trading right now, here are options available in to consider:
Why is Zinc Valuable?
Zinc is a bluish-white element that can be refined into a metal. It resists corrosion and, therefore, is often used to galvanize iron and steel.
Zinc is used to produce brass, which is a zinc alloy that contains between 55 and 95% copper.

Zinc’s History
Zinc was formally discovered as an element by Andreas Sigismund Marggraf, a German chemist who isolated it in 1746.
However, long before this discovery, civilizations were using zinc. According to the journal Ancient Asia, miners were smelting zinc in India by the ninth century BC.
During the Roman Empire, soldiers forged brass weapons from copper and zinc, and evidence from a smelting facility shows large-scale production of the metal took place in India between 1,100 and 1,500 AD.
Today mines worldwide extract more than 11.9 million metric tons of zinc annually. Zinc mines exist in more than 50 countries around the world, and the metal plays a key role in the steel industry.
Ready to start trading zinc? Read our trading guide.
Main Uses of Zinc
About one-third of zinc produced goes to galvanize other metals such as steel and iron. However, zinc has many other important applications.
| Uses | Description |
|---|---|
Galvanizing Material | Galvanization of steel and iron prevents corrosion and rusting. Electroplating is the most common method for galvanizing steel. Items including car bodies, street lamp posts, safety barriers and suspension bridges use galvanized steel. |
Alloys | Zinc combines with other metals to form strong alloys. Brass, nickel, silver, and aluminum solder are examples of zinc alloys. Musical instruments and hardware items use brass in construction, while electrical items and pipes use solder. |
Die-Castings | Die-casting is the production of metal parts using molds. The automobile, electrical and hardware industries all use zinc for die-casting parts. |
Zinc Oxide | Manufacturers of many products use zinc oxide in production, including paint, rubber, cosmetics, pharmaceuticals, plastics, inks, soaps, batteries, textiles, and electrical equipment. |
Zinc Sulfide | Zinc sulfide is an important component in many products, including luminous paints, fluorescent lights, and x-ray screens. |
Data last reviewed:
Zinc is also an essential nutrient for living things. A typical human body contains about 2.5 grams of zinc and consumes 15 milligrams per day. Foods with high zinc content include herring, beef, lamb, cheese, and sunflower seeds.
How is Zinc Produced?
The supply of zinc derives from two sources: primary production (mining) and secondary production (recycling). Mining provides most of the supply, although the United States Geological Survey (USGS) estimates the quantity recovered from recycling in the United States represented about 25% of total consumption.
Primary Production
More than 80% of zinc mining takes place in underground mines. Surface mining in open pits accounts for about 8% of mining, while mines that use both methods comprise the remaining 12%.
Several ores contain zinc including:
- zinc sulfide (sphalerite),
- zinc carbonate (smithsonite),
- zinc silicate (calamine), and
- manganese and iron compounds known as franklinite.

Zinc and lead ores usually occur together, and they often contain other valuable metals such as gold, silver and copper.
The process of primary production involves four steps:
- Ore concentration
- Smelting
- Refining
- Alloying
Ore Concentration
A series of steps called froth flotation breaks down the ores into particles with greater concentrations of zinc. First, the ore is ground into a fine powder and placed in flotation tanks with water, pine oil and chemicals
The tanks agitate the mixture and zinc particles float to the top, while clay and other silicates sink to the bottom.
The zinc particles are coated with chemicals to protect them from water. When air is injected into the tanks, the zinc particles adhere to the bubbles that form and remain at the top of the tank, while the remaining impurities float to the bottom. Scrapers then remove the bubbly mixture of concentrated zinc.
Filters remove the water and oils from the tank and leave behind a paste-like mixture. This mixture is combined with lime – the product made from heating limestone – and roasted in furnaces at temperatures of 2,500℉. This produces a solid mass of zinc oxide.
Smelting
During the second step, high-powered furnaces break down zinc oxide into elemental zinc.
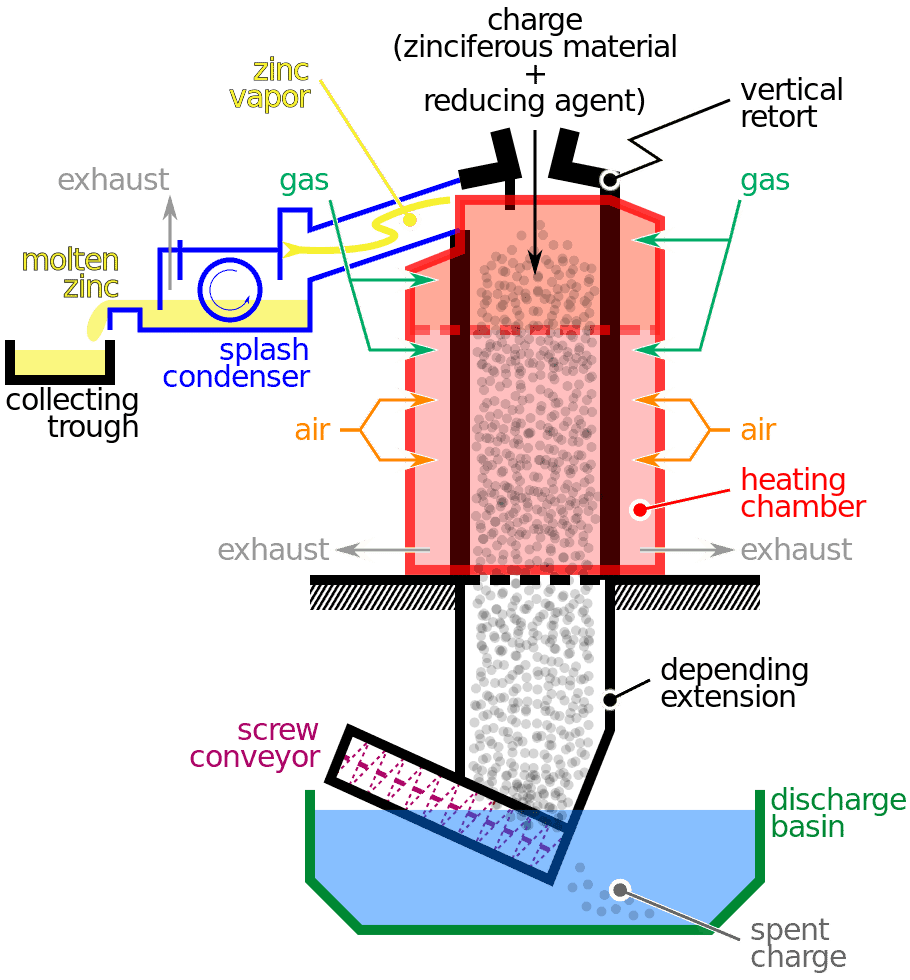
The furnaces use electricity, natural gas, or coal to reach temperatures of 2,200℉. A series of chemical reactions produces zinc and carbon dioxide. The carbon dioxide bonds with the zinc to re-form zinc oxide.
To keep the zinc separate, molten lead is introduced into the chamber. The zinc then dissolves into a molten state and gets carried to another chamber to cool.
Refining
The zinc molten, which is cooled to 824℉, can be further purified by cooling it in the chamber for several hours. During this refining stage, iron and other impurities sink to the bottom, while pure zinc remains at the top.
Alloying
Most zinc is alloyed with other metals. To make alloys, refiners re-melt zinc and combine it in exact proportions depending on the application.
Secondary Production
Recycling zinc is an economically viable activity. In the United States, about 35,000 tons of zinc is recycled annually. Recycling materials include residue from the process of galvanizing zinc with steel and zinc oxide recovered from blast furnaces.
Top Zinc Mining Countries

China is the leading zinc mining country in the world. It produces almost 40% of the annual supply of the metal.
| Rank | Flag | Country | Mine Production (Thousands of Metric Tons) |
|---|---|---|---|
| #1 |  | China | 4,500 |
| #2 |  | Peru | 1,300 |
| #3 |  | Australia | 850 |
| #4 |  | United States | 780 |
| #5 |  | Mexico | 710 |
| #6 |  | India | 650 |
| #7 |  | Bolivia | 460 |
| #8 |  | Kazakhstan | 340 |
| #9 |  | Canada | 310 |
| #10 |  | Sweden | 250 |
Data last reviewed:
Countries With the Largest Zinc Reserves
These are the reserves of each country as reported by the USGS:
| Rank | Flag | Country | Thousands of Metric Tons |
|---|---|---|---|
| #1 |  | Australia | 63,000 |
| #2 |  | China | 40,000 |
| #3 |  | Peru | 25,000 |
| #4 |  | Mexico | 17,000 |
| #5 |  | United States | 11,000 |
Data last reviewed: Warning: data published 5 years ago. Verify before relying on it.
What Drives the Price of Zinc?
The price of zinc is driven mostly by these five factors:
- Chinese Demand
- Chinese Supply
- Global Stocks
- US Demand
- Input Prices
Chinese Demand
As with most industrial commodities, China plays a pivotal role in determining zinc prices.
China is the top consumer of refined zinc used in galvanized steel.

Therefore, a key indicator of zinc demand in China and elsewhere is steel demand. Decisions about whether to undertake or hold off on infrastructure projects can create huge fluctuations in steel demand. Ultimately, these decisions can flow through to the zinc market.
Monetary policy from the People’s Bank of China is one important factor that can affect both steel and zinc demand. Stimulating measures can stoke demand for these commodities, while tighter monetary policies can depress demand.
China’s GDP growth has slowed considerably in recent years, creating doubts about future demand for all industrial metals including zinc. However, there are nascent signs that the economy may be rebounding.
Ultimately, zinc prices depend heavily on Chinese demand for galvanized steel in building projects.
Chinese Supply
A key factor impacting zinc output in China is the country’s increasing environmental awareness. Poor air quality has forced the government to take a harder look at the mining industry as a contributor to pollution.
If China curbs the production of zinc to deal with this problem, then the country will be more reliant on imports. This could drive prices higher.
Global Stocks
The London Metals Exchange (LME) keeps track of global stock levels for zinc and other industrial metals. Traders follow these inventory levels closely for clues about supply shortages or surpluses.
If inventory levels drop, the market may be facing a shortage of zinc supply in the near future. This could lead to higher prices for the metal. Similarly, if stockpiling occurs and inventory levels expand, then the market might face an oversupply of the metal, which can lead to lower prices.
One interesting development with base metals such as zinc is the increasing importance of inventories held at the Shanghai Futures Exchange (SHFE). Traders should monitor the change in zinc inventories at both exchanges for clues about zinc supply and prices.
US Demand
Construction and infrastructure represent a very large percentage of galvanized steel demand.
The United States has not invested in major infrastructure projects in decades. If the government earmarks funding for new infrastructure projects, the price for steel could move significantly higher. This would likely create increased demand for zinc and result in higher prices.
A strong US economy also increases demand for automobiles. Ultimately, the transportation sector is a very reliable barometer for galvanized steel and zinc demand.

Input Prices
Zinc occurs in ore bodies, and breaking down these ore bodies to extract the element expends energy. Mining and refining zinc requires ample supplies of coal, electricity and crude oil.
These costs can have a big effect on primary production. Similarly, the costs of scrap metal can impact the price of secondary production.
Where Can I Trade Zinc?
Start your research with reviews of these regulated brokers available in .
| Broker | Details | Sign up |
|---|---|---|
 | User friendly platform and leading risk management tools. | Open Account Now Plus500 Review |
| Social trading to copy leading traders. | Open Account Now eToro Review Copy Trading does not amount to investment advice. The value of your investments may go up or down. Your capital is at risk. |
|
 | No minimum deposit mobile platform with stocks and options. | Open Account Now Robinhood Review |
 | Options contracts trading fees from $2.09 on thousands of global stocks. | Open Account Now |
Data last reviewed:
CFDs are complex instruments and come with a high risk of losing money rapidly due to leverage. Between 50.00%-86.00% of retail investor accounts lose money when trading CFDs. You should consider whether you can afford to take the high risk of losing your money.
IMPORTANT: CFDs are not available in the USA.
Further Reading
Update history
This page was revised 4 times between September 2020 and March 2021.
Added introductory paragraph directing readers to zinc trading guides and broker options, simplified heading by removing "Regulated Brokers" language.
Added guide structure covering zinc production methods, global mining countries, price drivers, and nutritional importance with detailed explanations of primary and secondary production processes.
Added call-to-action content alert component in the How is Zinc Produced section.
Added new section detailing zinc's uses, historical discovery, mining methods, and production process with four subsections covering ore concentration through refining.
The Commodity Briefing
The stories behind the prices. Surprising, useful, occasionally weird - in your inbox every weekday.
- Price moves
- Supply shocks
- Macro drivers
Before you go
Get The Commodity Briefing - free, 2 minutes.